 Note how they are complete inverses of each other. I am attaching the images of the emission and absorption spectra of hydrogen atom. 
For absorption spectroscopy, varying wavelengths of photons are incident on the atom and the corresponding photon is absorbed by the atom resulting in no photon of that particular wavelength being detected. Below is the visible emission spectrum of hydrogen. The energy levels (n) are listed on the left side, followed by the energies of each level in J. The Paschen and Brackett series, with shorter arrows require the lower energy of the IR region. When atoms releases photons (light), the detector catches the production of the photon and gives out emission spectra. An energy level diagram for a hydrogen atom, not drawn to scale. What kind of spectrum you have at hand depends on which instrument and detector you are using. Papers dealing with the spectra of atoms with more than two electrons in the valence shell and continuous spectra of atoms are not included. When a photon is absorbed, the electron uses this energy to excite to a higher energy state. the atomic emission spectrum is the result of many excited atoms emitting light simultaneously. These observed spectral lines are due to the electron making transitions between two energy levels in an atom. The emission spectrum of atomic hydrogen has been divided into a number of spectral series, with wavelengths given by the Rydberg formula. Electromagnetic radiation also occurs as discreet packets called photons. 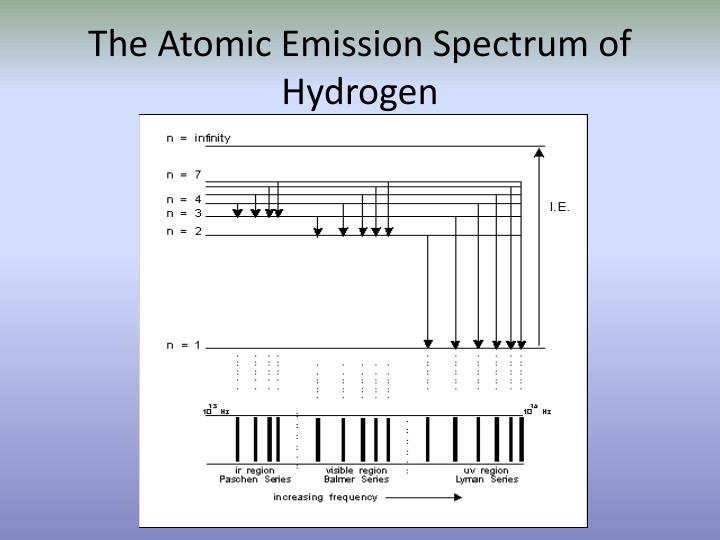
The relationship between the wavelength, frequency and speed of a wave is given by the equation: c × (1.11.1) (1.11.1) c ×. The hydrogen spectrum is complex, comprising more than the. The spectral series of hydrogen, on a logarithmic scale. All electromagnetic waves travel at the speed of light ( c c ), or 3.0 ×108m/s 3.0 × 10 8 m / s. However, the processes by which these two spectra are generated are completely the opposite of each other.Īn atom emits a photon when an electron goes from a higher energy state to a lower energy state, thereby releasing energy in the form of light. 12.3: The Atomic Spectrum of Hydrogen Extending hydrogens emission spectrum into the UV and IR. 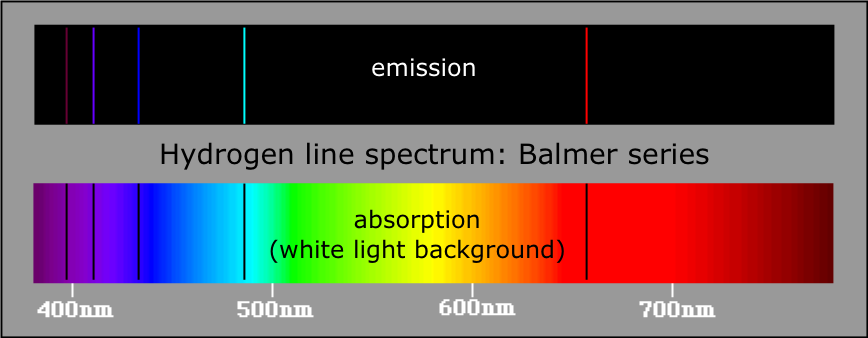
So, naturally, if an atom exhibits some emission spectrum, it will also give an absorption spectrum. Emission and absorption are inverse processes. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
